| Preliminary Clinical Study of Anti-HBV-DC Combine Lamivudine and Thymosin-A1 Treating the Chronic Hepatitis B Virus Carriers |
发布时间: 2010-04-02 人气指数:4275 |
The 20th Conference of the Asian Pacific Association for the Study of the Liver
Hepatology International.2010, 4(1):159
Bang-Fu Wu1,2, Jiang-Ying Yang2, Fang-Qin Li1, Wen-Bao Zhu1, Yun Zhou2, Fu-Xin Lin1, Yan-Ping Fu1, Jun Yang2, Chun-Qiong Hou1, Hui-Hua Zhou1, Wei Zheng1, Wei Chen1, Xue-Song Li1
2 Guangzhou Pubang Biological Immune Technology Research Center, Room 904, D District, Guangzhou International Business Incubator, Guangzhou Science City, Luogang District, Guangzhou, China
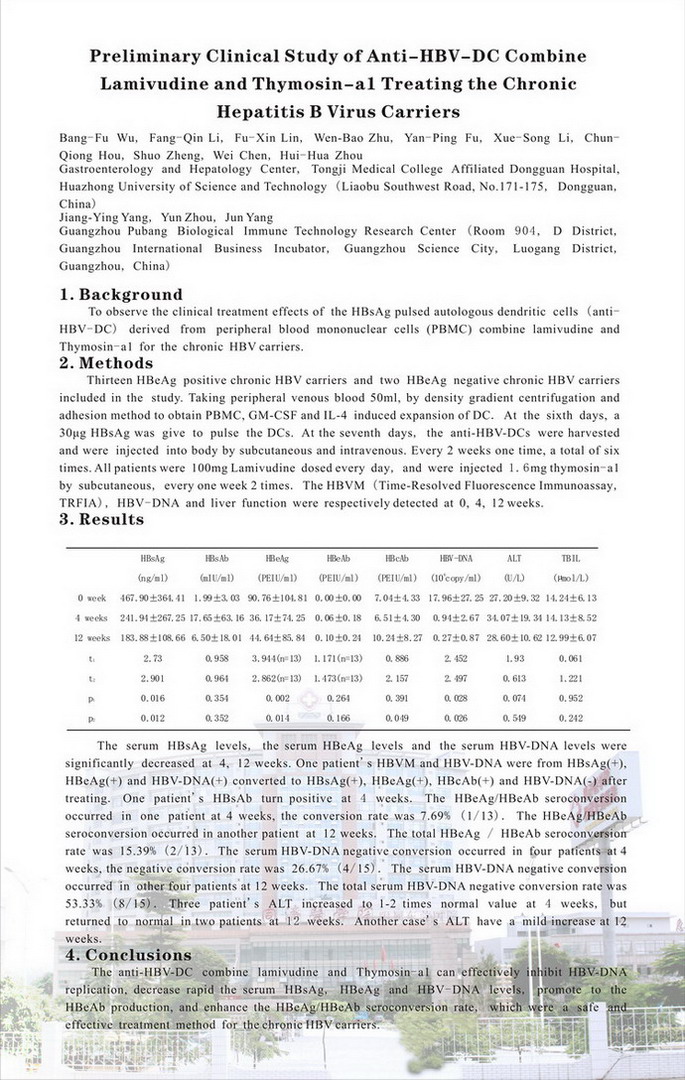
Background: To observe the clinical treatment effects of the HBsAg pulsed autologous dendritic cells (anti-HBV-DC) derived from peripheral blood mononuclear cells (PBMC) combine lamivudine and Thymosin-a1 for the chronic HBV carriers.
抗HBV-DC联合拉米夫定和胸腺肽a1治疗慢性HBV携带者的初步临床研究 第20届亚太肝脏研究会年会(北京) 海报展示(PP237) Hepatology International.2010, 4(1):159 华中科技大学同济医学院附属东莞医院消化肝病中心 吴邦富 李芳琴 凌佛鑫 朱文宝 付彦平 李雪松 侯春琼 郑硕 陈伟 周慧华 广州普邦生物免疫技术研究院 杨江英 周赟 杨军
目的 观察HBsAg致敏自体外周血单个核细胞(PBMC)来源的树突状细胞(抗HBV-DC)联合拉米夫定和胸腺肽a1治疗慢性HBV携带者的临床效果。 方法 HBeAg阳性慢性HBV携带者13人和HBeAg阴性慢性HBV携带者2人接受临床研究。取肝素抗凝外周静脉血50ml,以密度梯度离心及贴壁法获得PBMC,GM-CSF和IL-4诱导扩增DC,第6天给予30μg的HBsAg致敏DC,第7天收获抗HBV-DC,皮下和静脉各注射1/2。每2周1次,共6次。口服拉米夫定每次100mg,每天1次。皮下注射1.6mg胸腺肽a1,每周2次。分别于0、4、12周检测HBVM定量(时间分辨荧光免疫分析技术,TRFIA)、HBV-DNA定量及肝功能。 结果 0、4、12周的HBsAg分别为(467.90±364.41)ng/ml、(241.94±267.25)ng/ml(t=2.73,P=0.016)和(183.88±108.66)ng/ml(t=2.90,P=0.012);HBeAg(90.76±104.81)PEIU/ml、(36.17±74.25) PEIU/ml(t=3.94,P=0.002)和(44.64±85.84)PEIU/ml(t=2.86,P=0.014);HBV-DNA(17.96±27.25)×106copy/ml、(0.94±2.67)×106copy/ml(t=2.45,P=0.028)和(0.27±0.87)×106copy/ml(t=2.50,P=0.026)。治疗后4、12周的HBsAg、HBeAg和HBV-DNA显著下降。1例治疗前为HBsAg和HBeAg阳性,治疗后HBV-DNA显著下降,HBcAb转阳性。1例4周时HBsAb转阳性,未出现HBsAg/HBsAb的血清转换。1例4周时出现HBeAg/HBeAb血清转换,转换率7.69%(1/13),另1例12周时出现HBeAg/HBeAb血清转换,总转换率15.39%(2/13)。4例4周时HBV-DNA转阴,转阴率26.67%(4/15),另4例12周时HBV-DNA转阴,总HBV-DNA转阴率53.33%(8/15)。治疗前ALT均正常,治疗后3例4周时ALT升高1-2倍,其中2例12周时恢复正常,另1例12周时ALT轻度升高。 结论 抗HBV-DC联合拉米夫定和胸腺肽a1治疗,可有效地抑制慢性HBV携带者的HBV-DNA复制,快速降低血中HBsAg、HBeAg和HBV-DNA,促进HBeAb的产生,提高HBeAg/HBeAb的血清学转换率,是一种安全、有效的治疗慢性HBV携带者的方法。 |